

Here are graphs for some dim red light and some bright red light. nm (b) What is the lowest frequency of light incident on iron that releases photoelectrons from its surface Hz (c) If photons of energy 6. k is the wavelength of K luine which is a characteristic of an atom and does not depend on accelerating voltage of bombarding electron since k always refers to a photon wavelength of transition of e from the target element from.
That has a wavelength between 507.5 nm and 508.5 nm. Some related denitions are the cutoff frequency and the cutoff wavelength dened as follows: cckc,c 2 kc (cutoff frequency and wavelength) (9.1.9) We can then express in terms of and c,orin terms of and c. Thus, the cut -off wavelength is inversely proportional to acclerating voltage. The intensity of just that part of the light OptiFiber currently implements two different approaches for finding.
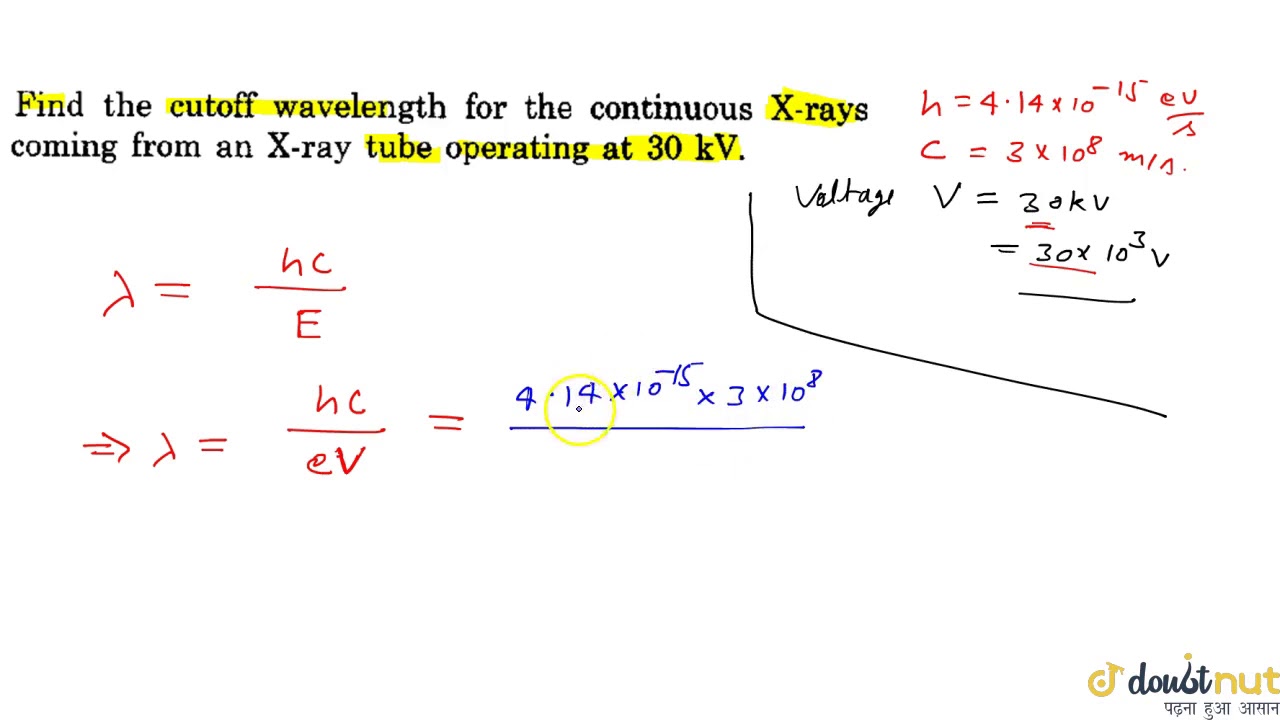
(What we display is intensity per unit wavelength, with units watts/m 2/nm. If we want to display information about how much energy is carriedĪt each wavelength, we can make a graph of intensity vs. The intensity of sunlight is roughly 1000 watts/m 2. Our book sometimes calls this energy flux, denoted with an F.īut why use a fancy word when a more well known word is available? Unit time is measured in watts, so intensity is measured in watts Is the amount of energy per unit time per unit area. Now, We also know that since the cut-off wavelength is given by h c e V If V is increased, will decrease and hence the cut-off point on the graph will shift leftwards. Above the cutoff wavelength, the fiber will only allow the LP01 mode to propagate through the. The Beer-Lambert Law, Equation \ref\): Three different types of cuvettes commonly used, with different usable wavelengths.We denote the energy content of light (or other electromagnetic The correct option is C As, the Kinetic energy of an electron is K e V 0, when we increase the accelerating potential of the circuit, the maximum kinetic energy of an electron increases. Cross section view of optical fiber and single fiber cable. The UV-vis region of energy for the electromagnetic spectrum covers 1.5 - 6.2 eV which relates to a wavelength range of 800 - 200 nm. What is actually being observed spectroscopically is the absorbance of light energy or electromagnetic radiation, which excites electrons from the ground state to the first singlet excited state of the compound or material. Ultraviolet-visible (UV-vis) spectroscopy is used to obtain the absorbance spectra of a compound in solution or as a solid.
